Vivli aims to advance human health through clinical research data sharing. One of the key ways we support this is through the Vivli platform, which facilitates data sharing for research teams working on a range of topics. These tables provide details of approved research proposals that have published or presented their results.
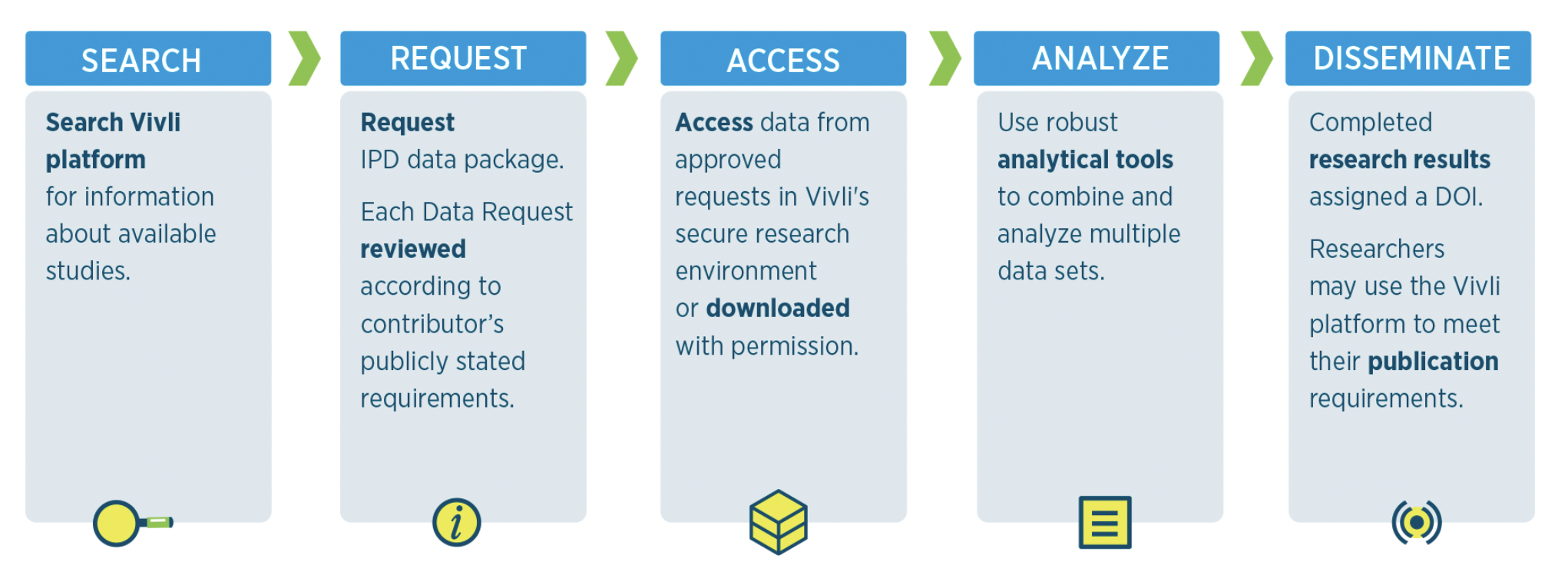
| Year | Data Request ID | Lead Investigator | Institution | Research Proposal Title | Publication | Data Contributor(s) | Altmetric Score | PubMed Citation Total |
|---|---|---|---|---|---|---|---|---|
| 2023 | 6104 | Pratik Sinha | Washington University | Heterogeneous treatment effects in molecular phenotypes of sepsis | Sinha, P., Kerchberger, V.E., Willmore, A., Chambers, J., Zhuo, H., Abbott, J., Jones, C., Wickersham, N., Wu, N., Neyton, L. and Langelier, C.R., 2023. Identifying molecular phenotypes in sepsis: an analysis of two prospective observational cohorts and secondary analysis of two randomised controlled trials. The Lancet Respiratory Medicine. Doi:10.1016/S2213-2600(23)00237-0 | Lilly | 81 | |
| 2021 | 5096 | Joo Sang Lee | Samsung Medical Center, South Korea | Harnessing genetic interactions to advance precision cancer medicine | Synthetic lethality-mediated precision oncology via the tumor transcriptome. Lee, Joo Sang et al. Cell, Volume 184, Issue 9, 2487 - 2502.e13. doi: 10.1016/j.cell.2021.03.030 | Tempus AI, Inc. | 69 | |
| 2023 | 4113 | Lesley Inker | Tufts Medical Center | Chronic Kidney Disease Epidemiology - Clinical Trials Consortium (CKD-EPI CT) | Inker, L.A., Collier, W., Greene, T., Miao, S., Chaudhari, J., Appel, G.B., Badve, S.V., Caravaca-Fontán, F., Del Vecchio, L., Floege, J. and Goicoechea, M., 2023. A meta-analysis of GFR slope as a surrogate endpoint for kidney failure. Nature Medicine, pp.1-10. Doi: 10.1038/s41591-023-02418-0 | GlaxoSmithKline, Takeda | 54 | |
| 2020 | 4540 | Chris Gale | University of Leeds | Efficacy and safety of edoxaban versus warfarin in patients with atrial fibrillation and frailty insights from the ENGAGE AF-TIMI 48 trial | Wilkinson, C., Wu, J., Searle, S.D. et al. Clinical outcomes in patients with atrial fibrillation and frailty: insights from the ENGAGE AF-TIMI 48 trial. BMC Med 18, 401 (2020). doi: 10.1186/s12916-020-01870-w | Daiichi Sankyo | 54 | |
| 2023 | 7875 | Marco Valgimigli | Cardiocentro Ticino, Lugano and Universita della Svizzera ltaliana (USI) | P2Y12 inhibitor or Aspirin moNoTHERapy as secondary prevention in patients with coronary artery disease: in individual patient data meta-analysis (PANTHER collaborative initiative) | Gragnano F, Cao D, Pirondini L, Franzone A, Kim HS, von Scheidt M, Pettersen AÃ…, Zhao Q, Woodward M, Chiarito M, McFadden EP, Valgimigli M. PANTHER. P2Y12 inhibitor or aspirin monotherapy for secondary prevention of coronary events. Journal of the American College of Cardiology. 2023 Jul 11;82(2):89-105. doi: 10.1016/j.jacc.2023.04.051 | Sanofi | 51 | |
| 2022 | 4116 | Sharon Strauss | St. Michael’s Hospital | Comparative safety and effectiveness of cognitive enhancers for Alzheimer's dementia a systematic review and individual patient data network meta-analysis | Veroniki AA, Ashoor HM, Rios P, et al. Comparative safety and efficacy of cognitive enhancers for Alzheimer’s dementia: a systematic review with individual patient data network meta-analysis BMJ Open 2022;12:e053012. doi: 10.1136/bmjopen-2021-053012 | AbbVie | 50 | |
| 2022 | 7074 | Gregory YH Lip | University of Liverpool | Impact of atrial fibrillation and the effects of comorbidities and treatment on disease progression and outcomes | Romiti GF, Proietti M, Bonini N, Ding WY, Boriani G, Huisman MV, Lip GYH; GLORIA-AF Investigators. Clinical Complexity Domains, Anticoagulation, and Outcomes in Patients with Atrial Fibrillation: A Report from the GLORIA-AF Registry Phase II and III. Thromb Haemost. 2022 Aug 29. doi: 10.1055/s-0042-1756355 | Boehringer Ingelheim | 50 | |
| 2020 | 4116 | Sharon Straus | St. Michael's Hospital | Comparative safety and effectiveness of cognitive enhancers for Alzheimer's dementia a systematic review and individual patient data network meta-analysis | Veroniki AA, Ashoor H, Rios P, Seitidis G, Mavridis D, Holroyd-Leduc J, Straus S, Tricco A. Comparative safety and efficacy of cognitive enhancers for Alzheimer’s dementia: An individual patient data network meta-analysis. In: Advances in Evidence Synthesis: special issue. Cochrane Database of Systematic Reviews 2020;(9 Suppl 1):455. doi: 10.1002/14651858.CD202001 | AbbVie | 50 | |
| 2020 | 5895, 6117 | Ashley Hopkins | Flinders University | Predictors of exposure, therapeutic and adverse effects of atezolizumab used in the treatment of advanced cancers | Concomitant Proton Pump Inhibitor Use and Survival in Urothelial Carcinoma Treated with Atezolizumab. Ashley M. Hopkins, Ganessan Kichenadasse, Christos S. Karapetis, Andrew Rowland and Michael J. Sorich. Clin Cancer Res October 15 2020 (26) (20) 5487-5493; doi: 10.1158/1078-0432.CCR-20-1876 | Roche | 49 | |
| 2022 | 4329 | Shomron Ben-Horin | Sun Yatsen 1st affiliated Hospital | Efficacy of biologic drugs in short-duration versus long-duration inflammatory bowel disease | Shomron Ben-Horin, Lena Novack, Ren Mao, Jing Guo, Yue Zhao, Ruslan Sergienko, Jian Zhang, Taku Kobayashi, Toshifumi Hibi, Yehuda Chowers, Laurent Peyrin-Biroulet, Jean Frederic Colombel, Gilaad G. Kaplan, Min-hu Chen. Efficacy of biologic drugs in short-duration versus long-duration inflammatory bowel disease: A systematic review and an individual-patient data meta-analysis of randomized controlled trials. Gastroenterology, 2021, ISSN 0016-5085. doi: 10.1053/j.gastro.2021.10.037 | AbbVie, Biogen, Johnson & Johnson, Takeda, UCB | 48 |
| Year | Data Request ID | Lead Investigator | Institution | Research Proposal Title | Publication | Data Contributor(s) | Altmetric Score | PubMed Citation Total |
|---|---|---|---|---|---|---|---|---|
| 2025 | 9813 | Sameer Jauhar | King's College London | Conducting a systematic review and meta-analysis to assess the incidence and severity of withdrawal symptoms following discontinuation of antidepressants | Kalfas, Michail, Tsapekos, Dimosthenis, Butler, Matthew, McCutcheon, Robert A., Pillinger, Toby, Strawbridge, Rebecca, Bhat, Bhagyashree Bhaskar, Haddad, Peter M., Cowen, Philip J., Howes, Oliver D., Joyce, Dan W., Nutt, David J., Baldwin, David S., Pariante, Carmine M., Lewis, Gemma, Young, Allan H., Lewis, Glyn, Hayes, Joseph F. and Jauhar, Sameer. "Incidence and Nature of Antidepressant Discontinuation Symptoms." JAMA Psychiatryvol. , no. , Jul. 2025, pp. https://doi.org/10.1001/jamapsychiatry.2025.1362 | Lilly, Lundbeck, Pfizer, Takeda | N/A | |
| 2023 | 6544 | Angela Wu | University of Oxford | Investigating the association between smoking cessation and mental health in people with and without psychiatric disorders | Wu, A.D., Gao, M., Aveyard, P. and Taylor, G., 2023. Smoking Cessation and Changes in Anxiety and Depression in Adults With and Without Psychiatric Disorders. JAMA Network Open, 6(5), pp.e2316111-e2316111. Doi: 10.1001/jamanetworkopen.2023.16111 | Pfizer | 17 | |
| 2025 | 5959 | John Dennis | University of Exeter Medical School | Stratification of SGLT2 inhibitor glucose lowering therapy in Type 2 diabetes | Dennis, J.M., Young, K.G., Cardoso, P., Güdemann, L.M., McGovern, A.P., Farmer, A., Holman, R.R., Sattar, N., McKinley, T.J., Pearson, E.R. and Jones, A.G., 2025. A five-drug class model using routinely available clinical features to optimise prescribing in type 2 diabetes: a prediction model development and validation study. The Lancet. Doi: 10.1016/S0140-6736(24)02617-5 | Boehringer Ingelheim, Takeda | N/A | |
| 2023 | 7875 | Marco Valgimigli | Cardiocentro Ticino, Lugano and Universita della Svizzera ltaliana (USI) | P2Y12 inhibitor or Aspirin moNoTHERapy as secondary prevention in patients with coronary artery disease: in individual patient data meta-analysis (PANTHER collaborative initiative) | Gragnano F, Cao D, Pirondini L, Franzone A, Kim HS, von Scheidt M, Pettersen AÃ…, Zhao Q, Woodward M, Chiarito M, McFadden EP, Valgimigli M. PANTHER. P2Y12 inhibitor or aspirin monotherapy for secondary prevention of coronary events. Journal of the American College of Cardiology. 2023 Jul 11;82(2):89-105. doi: 10.1016/j.jacc.2023.04.051 | Sanofi | 51 | |
| 2025 | 7810 | Diego Chowell | Icahn School of Medicine at Mount Sinai | Machine learning model to predict cancer immunotherapy response | Yoo, S.K., Fitzgerald, C.W., Cho, B.A., Fitzgerald, B.G., Han, C., Koh, E.S., Pandey, A., Sfreddo, H., Crowley, F., Korostin, M.R. and Debnath, N., 2025. Prediction of checkpoint inhibitor immunotherapy efficacy for cancer using routine blood tests and clinical data. Nature Medicine, pp.1-12. Doi: 10.1038/s41591-024-03398-5 | Roche | N/A | |
| 2025 | 9492, 8697 | David McAllister | University of Glasgow | Understanding frailty, multimorbidity and renal failure in clinical trials: Attrition, retention and heterogeneity of treatment effects in trials for diabetes, cancer and a heterogenous set of index conditions | Hanlon P., Butterly, E., Wei L. Age and Sex Differences in Efficacy of Treatments for Type 2 Diabetes. 2025. A Network Meta-Analysis. JAMA. Doi: 10.1001/jama.2024.27402 | AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Johnson & Johnson, Lilly, Roche, Sanofi, Takeda, UCB | ||
| 2023 | 7743 | Caroline Quach | CHU Sainte-Justine | Is there a Difference in the Immune Response, Efficacy, and Safety of Seasonal Influenza Vaccine in Males and Females? - A Meta-Analysis | Kiely, M., Tadount, F., Lo, E., Sadarangani, M., Wei, S.Q., Rafferty, E., Quach, C. and MacDonald, S.E., 2023. Sex differences in adverse events following seasonal influenza vaccines: a meta-analysis of randomised controlled trials. J Epidemiol Community Health. Doi: 10.1136/jech-2023-220781 | Sanofi | 4 | |
| 2022 | 5933, 5208 | Ahmad Abuhelwa | University of South Australia | Predictors of exposure, therapeutic and adverse effects of certolizumab pegol and baricitinib used in the treatment of rheumatoid arthritis | Daraghmeh DN, Hopkins AM, King C, Abuhelwa AY, Wechalekar MD, Proudman SM, Sorich MJ, Wiese MD. Female reproductive status and exogenous sex hormone use in rheumatoid arthritis patients treated with tocilizumab and csDMARDs. Rheumatology (Oxford). 2022 Jun 22:keac357. doi : 10.1093/rheumatology/keac357 | Roche | 2 | |
| 2023 | 5951, 4543 | Mathilde Nijkeuter | UMC Utrecht | Individualized prediction of recurrence risk reduction and risk of bleeding with extended anticoagulation in patients with venous thromboembolism | Maria A de Winter, Harry R Büller, Marc Carrier, Alexander T Cohen, John-Bjarne Hansen, Karin A H Kaasjager, Ajay K Kakkar, Saskia Middeldorp, Gary E Raskob, Henrik T Sørensen, Frank L J Visseren, Philip S Wells, Jannick A N Dorresteijn, Mathilde Nijkeuter, VTE-PREDICT study group. Recurrent venous thromboembolism and bleeding with extended anticoagulation: the VTE-PREDICT risk score. European Heart Journal, 2023;. ehac776. doi: 10.1093/eurheartj/ehac776 | Boehringer Ingelheim, Daiichi Sankyo | 18 | |
| 2023 | 7164 | Jörg Ellinger | University Hospital Bonn | Evaluation of CRP (c-reactive protein) flare phenomena as an early predictor of response to the Atezolizumab + Bevacizumab therapy in renal cell carcinoma | Saal, J., Bald, T., Eckstein, M., Ralser, D.J., Ritter, M., Brossart, P., Grünwald, V., Hölzel, M., Ellinger, J. and Klümper, N., 2023. Integrating On-Treatment Modified Glasgow Prognostic Score and Imaging to Predict Response and Outcomes in Metastatic Renal Cell Carcinoma. JAMA oncology. doi: 10.1001/jamaoncol.2023.1822 | Roche | 13 |


 Vivli’s mission is to promote, coordinate, and facilitate scientific sharing and reuse of clinical research data. As a neutral broker between data contributor, data user, and the wider data sharing community, Vivli maintains the trust of our community and clinical research by placing the utmost value on the security and safety of the data available within the Vivli Platform, a secure research environment (SRE).
Vivli’s mission is to promote, coordinate, and facilitate scientific sharing and reuse of clinical research data. As a neutral broker between data contributor, data user, and the wider data sharing community, Vivli maintains the trust of our community and clinical research by placing the utmost value on the security and safety of the data available within the Vivli Platform, a secure research environment (SRE).
 Dr. João Sérgio Neves is an endocrinologist, based in the Faculty of Medicine of the University of Porto and São João Hospital in Porto, Portugal. Dr. Neves’s team submitted a research proposal to access Vivli to conduct analysis relevant to their topic, “Albiglutide and Cardiovascular Outcomes in Type 2 Diabetes With and Without Concomitant Sodium-Glucose Cotransporter-2 Inhibition Use”. The team’s completed research has been presented in publications including the
Dr. João Sérgio Neves is an endocrinologist, based in the Faculty of Medicine of the University of Porto and São João Hospital in Porto, Portugal. Dr. Neves’s team submitted a research proposal to access Vivli to conduct analysis relevant to their topic, “Albiglutide and Cardiovascular Outcomes in Type 2 Diabetes With and Without Concomitant Sodium-Glucose Cotransporter-2 Inhibition Use”. The team’s completed research has been presented in publications including the  Did you know there’s more on Vivli than just clinical trial data? The majority of our repository of data comes from clinical trial data, but also includes significant numbers of platform trials, observational studies, and real-world evidence resources.
Did you know there’s more on Vivli than just clinical trial data? The majority of our repository of data comes from clinical trial data, but also includes significant numbers of platform trials, observational studies, and real-world evidence resources.